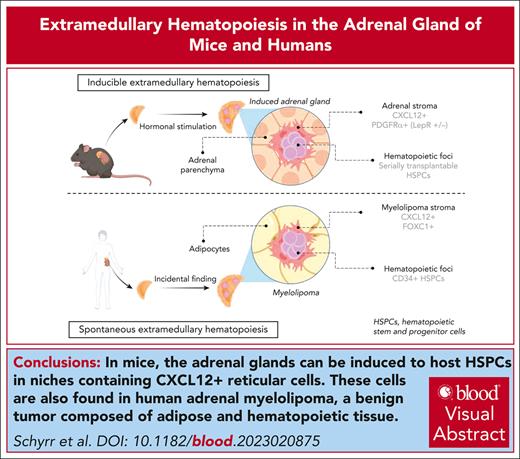
Key Points
The adrenal gland can be hormonally induced to host serially transplantable HSPCs in adult mice.
Adrenal extramedullary hematopoiesis is associated to the formation of PDGFRα+LEPR+/− foci in mice and CXCL12+FOXC1+ stroma in humans.
Visual Abstract
Adult hematopoietic stem and progenitor cells (HSPCs) reside in the bone marrow (BM) hematopoietic niche, which regulates HSPC quiescence, self-renewal, and commitment in a demand-adapted manner. Although the complex BM niche is responsible for adult hematopoiesis, evidence exists for simpler, albeit functional and more accessible, extramedullary hematopoietic niches. Inspired by the anecdotal description of retroperitoneal hematopoietic masses occurring at higher frequency upon hormonal dysregulation within the adrenal gland, we hypothesized that the adult adrenal gland could be induced into a hematopoietic-supportive environment in a systematic manner, thus revealing mechanisms underlying de novo niche formation in the adult. Here, we show that upon splenectomy and hormonal stimulation, the adult adrenal gland of mice can be induced to recruit and host functional HSPCs, capable of serial transplantation, and that this phenomenon is associated with de novo formation of platelet-derived growth factor receptor α/leptin receptor (PDGFRα+/LEPR+/–)–expressing stromal nodules. We further show in CXCL12–green fluorescent protein reporter mice that adrenal glands contain a stromal population reminiscent of the CXCL12-abundant reticular cells, which compose the BM HSPC niche. Mechanistically, HSPC homing to hormonally induced adrenal glands was found dependent on the CXCR4–CXCL12 axis. Mirroring our findings in mice, we found reticular CXCL12+ cells coexpressing master niche regulator FOXC1 in primary samples from human adrenal myelolipomas, a benign tumor composed of adipose and hematopoietic tissue. Our findings reignite long-standing questions regarding hormonal regulation of hematopoiesis and provide a novel model to facilitate the study of adult-specific inducible hematopoietic niches, which may pave the way to therapeutic applications.
Introduction
Although adult hematopoiesis takes place primarily in the bone marrow (BM), examples of adult hematopoiesis outside the bone cavity exist, grouped under the term extramedullary hematopoiesis (EMH). EMH can be classified in 2 groups1: (1) EMH arising in fetal hematopoiesis sites, fundamentally the spleen and liver; and (2) adult-specific EMH in nonfetal hematopoietic sites.
EMH in nonfetal hematopoietic sites can arise either spontaneously as a benign tumor, possibly driven by a stromal population,2,3 or upon extreme hematopoietic demand.4 Benign EMH masses in nonfetal hematopoietic sites occur with a particular high frequency in the adrenal gland, constituting a distinct clinical entity called adrenal myelolipoma, with a prevalence at autopsy estimated to be between ∼0.08% and 0.2%.3 Myelolipomas are disproportionally associated with congenital adrenal hyperplasia (with a prevalence of up to 6%5) a condition that associates high circulating adrenocorticotropic hormone (ACTH) levels.6
We hypothesized that exogenous hormonal stimulation may induce a hematopoietic niche in the adrenal glands. We show that the murine adrenal gland can be induced by ACTH to form a hematopoietic-supportive tissue and used as a model to study the components of a minimalistic adult hematopoietic niche, bypassing the need for an ossified structure. EMH-induced adrenal glands contain serially transplantable hematopoietic stem and progenitor cells (HSPCs), host HSPCs upon adrenal induction within newly formed platelet-derived growth factor receptor α (PDGFRα+) stromal niches and retain CD45+ hematopoietic cells in a CXCR4-dependent manner. CXCL12–green fluorescent protein (GFP) reporter mice reveal numerous CXCL12+ stromal cells with reticular morphology within the adrenal gland resembling CXCL12-abundant reticular (CAR) cells. Furthermore, we show that human myelolipoma samples also contain an abundant CXCL12+ population, mirroring the findings of our murine model.
Methods
Detailed experimental methods are provided in supplemental Methods, available on the Blood website.
Surgery
For splenectomy, animals were placed on their right flank. The spleen was exposed, and the splenic vessels were ligated. Then, the spleen was removed. A minimal period of 7 days was observed before starting any experiment.
EMH induction cocktail and inhibitors
For induction of EMH in the adrenal gland, mice were injected daily subcutaneously for 20 to 21 days with granulocyte colony-stimulating factor (G-CSF; 150 μg/kg filgrastim 30 mio U/0.5 mL corresponding to 300 μg/0.5mL [Neupogen; Amgen], diluted in Glucosum [Bichsel] solution 5 % or NaCl 0.9% as vehicle), testosterone (310 μg per mouse, testosterone undecanoate 1000 mg/4 mL [Nebido; Bayer] diluted in corn oil [C8267-500mL; Sigma-Aldrich] for a final volume of 30 μL, subcutaneously) and ACTH (tetracosactide, 20 μg per mouse subcutaneously [Synacthen Depot solution for injection, 1 mg/mL; Alfasigma], combined with either the testosterone or corn oil vehicle).
Colony-forming unit assay (CFU)
For CFU assays from BM cells, 10 000 CD45+ were plated in methylcellulose and assessed upon 8 days of culture.
Homing assay CXCR4i
After EMH induction in wild-type C57BL/6J recipient mice, we used fluorescence-activated cell sorting to sort 35 000 GFP+ lineage– c-Kit+ Sca-1+ (LKS) cells (B6.ACTB-GFP) per recipient mouse into phosphate-buffered saline plus 2% fetal bovine serum. We incubated the GFP+ LKS cells with plerixafor 1 μM (AMD3100, Selleckchem) and injected them intravenously.7 At 30 minutes, and 12 and 24 hours after LKS injection we administered plerixafor dissolved in phosphate-buffered saline (10 mg/kg) intraperitoneally to recipient mice, as previously described.8 The adrenal glands were collected at 36 hours after LKS injection.
Histology
For immunohistochemistry (IHC), detection was performed manually with 3,3’-diaminobenzidine (D5905, Sigma-Aldrich) or Discovery purple/Discovery teal. Sections were counterstained with Harris or Mayer hematoxylin.
Immunofluorescence and confocal microscopy
Excised adrenal glands were cut to 200-μm-thick whole-mount sections, that were imaged with a Leica STELLARIS V.
For adrenal sections, optimal cutting temperature–embedded organs were cut into 5-μm-thick sections and imaged with a Nikon Ti2 spinning-disk confocal microscope. All antibodies used can be found in Table 1 and supplemental Methods.
Semiquantitative analysis of immunohistochemical stains on human myelolipoma samples
| Sample ID . | CD34 . | CD73 . | CD90 . | CD146 . | CD271 . | CXCL12 . | NESTIN . | Anatomical origin . |
|---|---|---|---|---|---|---|---|---|
| A | + | / | +++ | +++ | / | ++ | +++ | Adrenal |
| B | - | + | - | ++ | + | + | ++ | Retroperitoneum |
| C | + | + | + | +++ | +++ | ++ | ++ | Pelvic |
| D | - | + | + | ++ | +++ | +++ | + | Adrenal |
| E | - | +++ | +++ | +++ | +++ | + | +++ | Adrenal |
| F | - | ++ | ++ | +++ | +++ | + | +++ | Adrenal |
| G | - | ++ | + | ++ | +++ | - | +++ | Adrenal (adenoma) |
| H | - | ++ | + | ++ | +++ | + | +++ | Adrenal |
| I | + | +++ | + | ++ | +++ | + | ++ | Pelvic |
| J | - | + | + | + | ++ | - | + | BM |
| K | + | + | ++ | ++ | +++ | + | +++ | Adrenal |
| Sample ID . | CD34 . | CD73 . | CD90 . | CD146 . | CD271 . | CXCL12 . | NESTIN . | Anatomical origin . |
|---|---|---|---|---|---|---|---|---|
| A | + | / | +++ | +++ | / | ++ | +++ | Adrenal |
| B | - | + | - | ++ | + | + | ++ | Retroperitoneum |
| C | + | + | + | +++ | +++ | ++ | ++ | Pelvic |
| D | - | + | + | ++ | +++ | +++ | + | Adrenal |
| E | - | +++ | +++ | +++ | +++ | + | +++ | Adrenal |
| F | - | ++ | ++ | +++ | +++ | + | +++ | Adrenal |
| G | - | ++ | + | ++ | +++ | - | +++ | Adrenal (adenoma) |
| H | - | ++ | + | ++ | +++ | + | +++ | Adrenal |
| I | + | +++ | + | ++ | +++ | + | ++ | Pelvic |
| J | - | + | + | + | ++ | - | + | BM |
| K | + | + | ++ | ++ | +++ | + | +++ | Adrenal |
Detailed semiquantitative histological characteristics of the different immunohistochemical stains on the myelolipoma samples and 2 controls (healthy BM [J] and adrenal adenoma [G]), quantification represent number of positive cells per samples (“-” corresponds to no positive cell; “+” to “+++” correspond to linearly increasing number of positive cells; and “/” corresponds to no data).
Quantification and statistics
P values were calculated using unpaired 2-tailed Student t test or 1-way analysis of variance with GraphPad Prism 9; ∗P < .05, ∗∗P < .01, ∗∗∗P < .001, and ∗∗∗∗P < .0001.
For human data, all procedures were in accordance with the ethical standards of the responsible committee on human experimentation and in accordance with the 1975 Declaration of Helsinki as revised in 2008. The Comission cantonale d'étique de la recherche sur sur l'être humain (CER-VD; Lausanne, Switzerland) local ethical commission approved the study and a specific consent for this study was obtained in cases in which a general consent for research was not already available. For cases in which the effort to obtain a specific consent were disproportionate, specific consent was waived by the CER-VD according to the provision of the Swiss Federal Human Research Ordinance (RS 810.301). For animal studies, experiments were carried out in accordance with the Swiss law and with approval of the cantonal authorities (Service Vétérinaire de l’Etat de Vaud) and ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines.
Results
The adrenal gland can be hormonally induced to host hematopoietic cells
The description of adrenal myelolipomas as boneless EMH masses in several mammalian species prompted us to hypothesize that the adult adrenal gland could be induced into an adult-specific hematopoietic-supportive environment. Selye and Stone, in 1950, described the possibility of transforming the adrenal gland into myeloid-like tissue through stimulation with pituitary gland extracts, testosterone, and tumor lysates.9
Based on this model, we designed a strategy to induce EMH in the murine adrenal gland. We injected a hematopoietic cytokine (G-CSF) and the pituitary axis adrenocorticotropic hormone (ACTH) as well as an androgen (testosterone undecanoate) in splenectomized mice (Figure 1A). Mice developed symptoms of hypercortisolism (Cushing syndrome) within the first 7 days of injection, showing increased weight, polyuria, and polydipsia10 (supplemental Figure 1A-B). EMH induction treatment modestly increased circulatory white blood cells and granulocytes, as expected upon G-CSF administration, but had no effect on hemoglobin (supplemental Figure 1C-E).
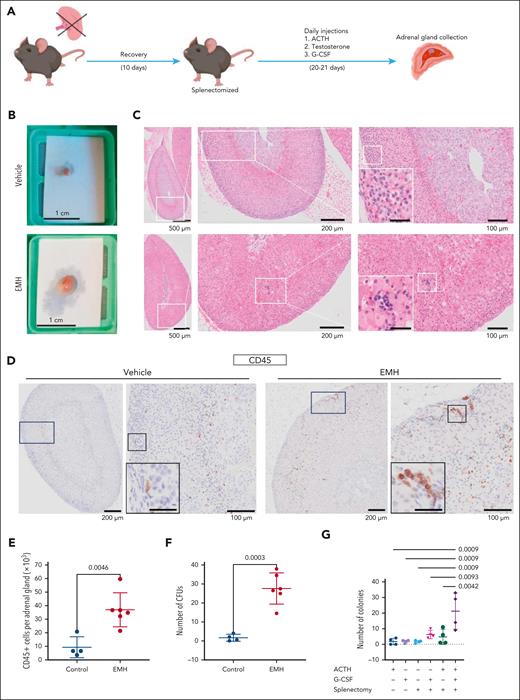
The adrenal gland can be hormonally induced to host hematopoietic cells. (A) General experimental design to induce EMH in the adrenal gland. (B) Macroscopic picture of freshly isolated adrenal glands. (C) Representative images of hematoxylin and eosin (H&E) stains for vehicle (top) and EMH-induced (bottom) adrenal glands. The boxed area is magnified on the right of each image. Scale bar for inset represents 30 μm. (D) Representative images of CD45 IHC stain in an adrenal gland. The boxed area is magnified on the right of each image. Scale bar for inset represents 30 μm. (E) Number of CD45+ cells per adrenal gland, measured by flow cytometry (control, n = 4; EMH, n = 6 mice; 2 independent experiments). (F) CFU assay, number of colonies per adrenal gland (control, n = 4; EMH, n = 6 mice; 2 independent experiments). (G) Number of hematopoietic colonies per adrenal gland obtained in a CFU assay from mice treated with the different components of the induction protocol (n = 4 for all groups). Data are represented as mean ± standard deviation (SD). Differences were assessed using unpaired, 2-tailed Student t test (E-F) or with 1-way analysis of variance (ANOVA) followed by Holms-Šidák multiple correction test (G). P values are indicated in the graphs.
The adrenal gland can be hormonally induced to host hematopoietic cells. (A) General experimental design to induce EMH in the adrenal gland. (B) Macroscopic picture of freshly isolated adrenal glands. (C) Representative images of hematoxylin and eosin (H&E) stains for vehicle (top) and EMH-induced (bottom) adrenal glands. The boxed area is magnified on the right of each image. Scale bar for inset represents 30 μm. (D) Representative images of CD45 IHC stain in an adrenal gland. The boxed area is magnified on the right of each image. Scale bar for inset represents 30 μm. (E) Number of CD45+ cells per adrenal gland, measured by flow cytometry (control, n = 4; EMH, n = 6 mice; 2 independent experiments). (F) CFU assay, number of colonies per adrenal gland (control, n = 4; EMH, n = 6 mice; 2 independent experiments). (G) Number of hematopoietic colonies per adrenal gland obtained in a CFU assay from mice treated with the different components of the induction protocol (n = 4 for all groups). Data are represented as mean ± standard deviation (SD). Differences were assessed using unpaired, 2-tailed Student t test (E-F) or with 1-way analysis of variance (ANOVA) followed by Holms-Šidák multiple correction test (G). P values are indicated in the graphs.
Adrenal glands from induced mice were markedly larger than those from the control group (Figure 1B). Upon hematoxylin and eosin histological examination, foci of hematopoietic cells could be identified morphologically in the adrenal cortex of EMH-induced mice (Figure 1C) and further confirmed with IHC for CD45, a panhematopoietic marker (Figure 1D). EMH was not detected in the kidney, pancreas, ovary, white adipose tissue, brown adipose tissue, or omentum. We did find, however, foci in the liver, which are congruent with the described induction of EMH in this organ upon G-CSF treatment.11 These foci were morphologically similar to those found in the adrenal glands, and contained small, basophilic cells with a low cytoplasm-to-nucleus ratio (supplemental Figure 1F). We found in the adrenal cells positive for von Willebrand factor, a marker of megakaryocytes, suggesting in situ hematopoiesis (supplemental Figure 1G). Because EMH foci contain small cells that could resemble lymphocytes, we performed IHC for CD3 and B220 in the adrenal glands to rule out a lymphocytic infiltrate. Both markers were predominantly negative in the EMH foci of the adrenal glands (supplemental Figure 1H). Conversely, we found Ter119-positive cells in the EMH foci of the induced adrenal glands, indicating nucleated cells of erythroid identity (supplemental Figure 1H). Although Selye and Stone9 mentioned the presence of adipocytes in the induced adrenal glands of their rat model, we could not detect mature adipocytes in our samples. The increased number of hematopoietic cells in the induced adrenal glands was quantified by flow cytometry (Figure 1E), which revealed a fourfold increase in CD45+ cells in the glands retrieved from the treated group.
CFU assays measure the progenitor function of short-term HSPCs.12 This assay showed that cells within the induced adrenal glands form more colonies than those obtained from the control glands, accounting for a 15-fold increase in total colonies (Figure 1F). The increase in CFUs was statistically significant also when normalized to the total CD45+ count to take into consideration the increased adrenal volume and thus higher numbers of CD45+ cells in the induced glands (supplemental Figure 1I). Both the induction cocktail and splenectomy were necessary for the full development of the phenotype (Figure 1G; supplemental Figure 1J). Collectively, these results indicate that the adrenal glands can be hormonally induced to selectively enrich in hematopoietic cells with increased colony-forming potential.
The induced adrenal gland contains functional, serially transplantable HSPCs
Once we had determined the presence of hematopoietic cells with CFU potential, we investigated the nature of the hematopoietic cells found in the induced adrenal glands. For this we performed flow cytometry for known HSPC surface markers. Within the BM, the immunophenotype of HSPCs, defined as Lineagelowc-Kit+Sca1+ (LKS), showed no difference between control and induced mice (Figure 2A). In the blood, the induction cocktail caused an increase in circulating c-Kit+ progenitor cells, as expected, due to G-CSF administration. In the adrenal gland, a different surface marker profile was observed. Specifically, CD45+ lineage cells obtained from the induced adrenal glands did not show the same surface marker profile as in the BM but instead a proportion of them displayed a c-Kitlow and Sca-1+ profile (Figure 2A). This is congruent with previous reports that have shown a decrease in the expression of c-Kit in splenic hematopoietic stem cells (HSCs).13 We then interrogated the cells in this gate for the presence of the SLAM markers CD150 and CD48,14 which are used to identify multipotent progenitors (LKS CD150–CD48+/−) and HSCs (LKS CD150+CD48–) in the murine BM. In doing so, we observed both LKSlow CD150+CD48– events, and thus, hematopoietic stem-like cells, and LKSlow CD150+CD48+ or CD150–CD48+/− multipotent progenitor-like cells (supplemental Figure 2A). This immunophenotypical signature suggests that HSPCs are present in the EMH-induced adrenal gland. However, because of the limited number of hematopoietic cells in the adrenal glands and the seemingly lower expression of their markers, cell sorting could not be performed to investigate the functional capacities of the lineage populations present in the induced adrenal glands. Instead, we performed functional transplantation assays.
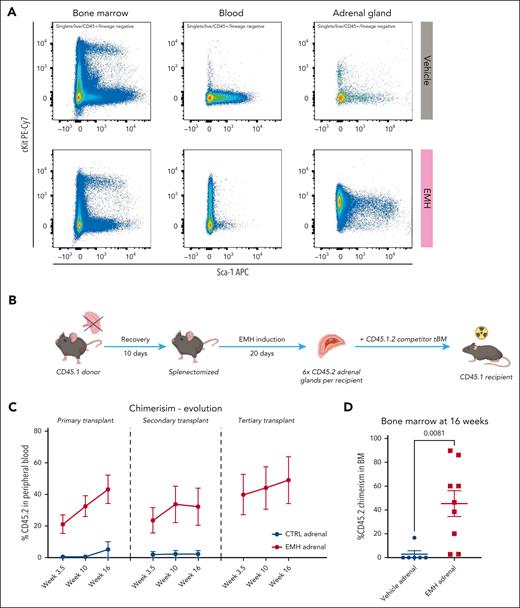
The adrenal gland supports functional, serially transplantable HSPCs. (A) Flow cytometry analysis of the BM, blood, and adrenal glands of control vs EMH-treated mice; representative panels gated within the CD45+ Lin− gate are shown (control, n = 10; EMH, n = 10 mice; 3 independent experiments). (B) Experimental design of the competitive transplant. The total content of CD45.2+ cells retrieved from 6 adrenal glands were transplanted into a lethally irradiated CD45.1 recipient together with 125 000 CD45.1.2 total BM cells from a competitor mouse. (C-D) Evolution of the CD45.2 donor engraftment in serial transplantation as measured in the peripheral blood and BM, respectively, by flow cytometry (control, n = 7; EMH, n = 8 mice; 2 independent experiments). Data are shown as mean ± standard error of the mean. Differences were assessed using a 2-tailed unpaired Student t test.
The adrenal gland supports functional, serially transplantable HSPCs. (A) Flow cytometry analysis of the BM, blood, and adrenal glands of control vs EMH-treated mice; representative panels gated within the CD45+ Lin− gate are shown (control, n = 10; EMH, n = 10 mice; 3 independent experiments). (B) Experimental design of the competitive transplant. The total content of CD45.2+ cells retrieved from 6 adrenal glands were transplanted into a lethally irradiated CD45.1 recipient together with 125 000 CD45.1.2 total BM cells from a competitor mouse. (C-D) Evolution of the CD45.2 donor engraftment in serial transplantation as measured in the peripheral blood and BM, respectively, by flow cytometry (control, n = 7; EMH, n = 8 mice; 2 independent experiments). Data are shown as mean ± standard error of the mean. Differences were assessed using a 2-tailed unpaired Student t test.
CD45.2 donor mice were thus treated with the EMH induction cocktail, and the cells obtained by enzymatic digestion of the adrenal glands were directly transplanted together with CD45.1/.2 BM competitor cells into lethally irradiated CD45.1 recipient mice. Based on the CFU data (Figure 1F), 6 adrenal glands contain a similar colony-forming potential as 125 000 total BM cells, the minimal BM cell rescue dose in our experimental setup. Therefore, we transplanted the total cellular content of 6 adrenal glands-control or EMH-induced-together with 125 000 total BM competitor cells for a 1:1 competitive transplant assay. CD45.2+ cells in the blood of the CD45.1 primary recipient indicate engraftment originating from our donor mice and thus reveal adrenal resident HSPCs (Figure 2B). We observed significant CD45.2 engraftment exclusively for mice receiving CD45.2 donor cells from EMH-induced adrenal glands but not from control adrenal glands (Figure 2C). Donor cells gave rise to both myeloid and lymphoid circulating blood cells (supplemental Figure 2B). More importantly, CD45.2 cells obtained from EMH-induced adrenal glands were serially transplantable and capable of producing circulating cells up to at least tertiary transplants (Figure 2C). Long-term CD45.2 engraftment was also observable in the BM of mice receiving cells from EMH-induced donors, thereby demonstrating the presence of functional HSPCs capable of homing to the endogenous BM in the EMH-induced adrenal glands, but not the uninduced controls (Figure 2D).
Induced adrenal glands recruit circulating HSPCs
After identifying the hematopoietic supporting capacity of the induced adrenal glands, we sought to define the source of the observed HSPCs. Embryologically, the aorta-gonad-mesonephros (AGM) structure gives rise to both the definitive HSPCs and the adrenal cortex. During embryonic development, in the AGM, HSPCs derive from endothelial cells in an endothelial-to-hematopoietic transition.15 Even if unlikely, we wanted to exclude that the HSPCs we observed in the adrenal glands could develop in situ from nonhematopoietic adrenal cells upon EMH induction. To test this hypothesis, we transplanted splenectomized mice with GFP+ total BM cells to obtain a mouse with a GFP+ hematopoietic system. After recovery from transplantation, mice were treated with the EMH induction cocktail for 20 days (Figure 3A). If direct metaplasia occurred, adrenal HSPCs would be GFP– upon EMH-induction, whereas BM HSPCs would be GFP+. As in previous assays, CFU assays showed the presence of hematopoietic progenitors in the induced adrenal glands (Figure 3B) but not in control mice, as well as in the BM of all mice (Figure 3C). It should be noted that the baseline number of colonies was reduced by ∼50% in irradiated mice as compared with all other experiments performed in nonirradiated EMH-induced adrenal glands. In these conditions, we found that close to 100% of all colonies present in the adrenal gland CFU assays were GFP+, indicating a BM origin of the hematopoietic cells in the adrenal gland upon EMH induction. Overall, these results indicate that HSPCs found in the adrenal gland are recruited from the BM into the induced adrenal gland and do not arise de novo in the organ.
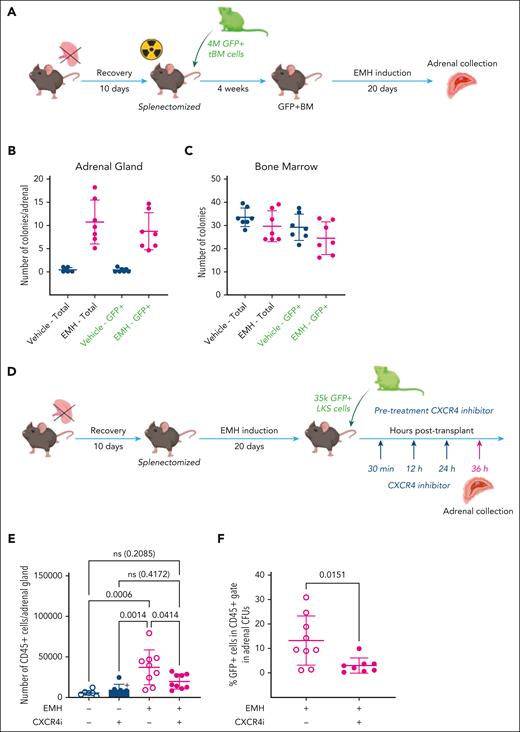
The adrenal stroma recruits and supports circulating hematopoietic progenitors. (A) Experimental design for EMH induction after transplant with GFP+ BM. (B-C) CFU assay, number of total colonies (left, black labels) and GFP+ colonies (right, green labels) from total per single adrenal gland (B) and 10 000 CD45+ total BM cells (C); n = 6 per experimental group, 2 independent experiments). (D) General experimental design for homing assay. (E) CD45+ cells in EMH-induced adrenals glands retrieved from mice treated with plerixafor (CXCR4i), evaluated by flow cytometry at 16 hours after treatment (2 independent experiments, n = 8 for control groups, and n = 10 for EMH-induced groups). (F) Total cells were recovered after 8-day CFU assay in complete methylcellulose and the percentage of GFP+ cells from the recovered cells is shown, as quantified by flow cytometry (n = 9 control mice, and n = 8 experimental mice). Data are represented as mean ± SD and groups were compared with 1-way ANOVA followed by Tukey multiple correction test (E) or 2-tailed, unpaired Student t test (F). ns, not significant.
The adrenal stroma recruits and supports circulating hematopoietic progenitors. (A) Experimental design for EMH induction after transplant with GFP+ BM. (B-C) CFU assay, number of total colonies (left, black labels) and GFP+ colonies (right, green labels) from total per single adrenal gland (B) and 10 000 CD45+ total BM cells (C); n = 6 per experimental group, 2 independent experiments). (D) General experimental design for homing assay. (E) CD45+ cells in EMH-induced adrenals glands retrieved from mice treated with plerixafor (CXCR4i), evaluated by flow cytometry at 16 hours after treatment (2 independent experiments, n = 8 for control groups, and n = 10 for EMH-induced groups). (F) Total cells were recovered after 8-day CFU assay in complete methylcellulose and the percentage of GFP+ cells from the recovered cells is shown, as quantified by flow cytometry (n = 9 control mice, and n = 8 experimental mice). Data are represented as mean ± SD and groups were compared with 1-way ANOVA followed by Tukey multiple correction test (E) or 2-tailed, unpaired Student t test (F). ns, not significant.
CXCL12 is required for homing and retention of hematopoietic cells in the induced adrenal gland
Once we had determined that the induced adrenal gland can be colonized by hematopoietic cells with CFU potential originating from the BM, we hypothesized that the CXCL12–CXCR4 axis would be involved in this phenomenon, given its crucial role in homing of hematopoietic progenitor cells to the BM.8 To evaluate this hypothesis, we used plerixafor, a pharmacological antagonist of CXCR4. We performed the EMH-induction protocol and then injected the EMH-induced mice with 35 000 GFP+ LKS cells treated with plerixafor. We administered plerixafor intraperitoneally at 30 minutes, 12 hours, and 24 hours after LKS injection.8 The mice were euthanized 36 hours after LKS injection and the adrenal glands evaluated for CD45+ counts and GFP+ CFU potential (Figure 3D-E). We expected plerixafor to hamper the colonization of the adrenal niche by the injected GFP+ LKS cells, and therefore a decrease in the number of GFP+ cells produced in the CFU assay. Congruently, upon EMH induction, we observed a marked decrease in the number of CD45+ cells in the adrenal glands of induced mice treated with plerixafor (Figure 3E), indicating that the CXCL12–CXCR4 axis is necessary for the retention of CD45+ cells in the adrenal niche. We then evaluated by flow cytometry the proportion of GFP+ cells within the colonies. Consistent with our hypothesis, we observed a decrease in the percentage of GFP+ cells that composed the hematopoietic colonies in EMH-induced animals treated with plerixafor (Figure 3F), indicating that the CXCL12–CXCR4 axis is required not just for the retention but also for the homing of hematopoietic cells with CFU potential to the adrenal gland.
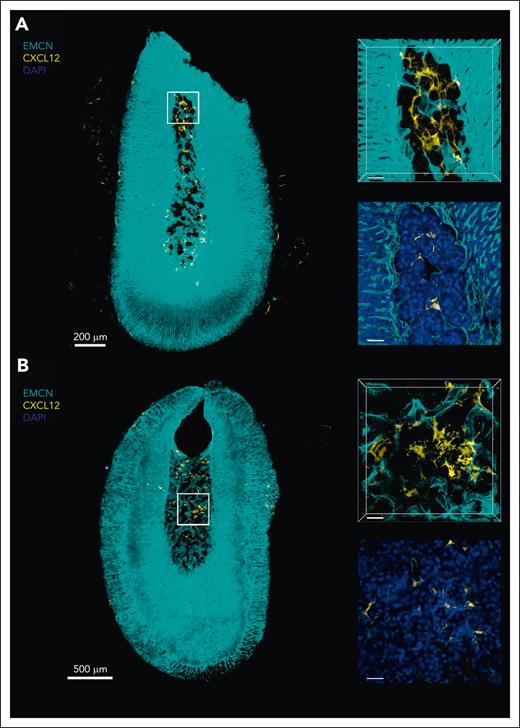
Finally, we looked into the presence of CAR cells in the adrenal gland, which have been described to be essential for hematopoietic support and retention in the BM.16 For this, we took advantage of the extensively characterized CXCL12-GFP knockin murine reporter model.17 We identified GFP+ cells in both the induced and noninduced adrenal gland by flow cytometry (data not shown) and confirmed our findings with whole-mount confocal microscopy (Figure 4A-B control; EMH induced). These cells were of reticular morphology, reminiscent of CAR cell morphology in the BM.18 Surprisingly, and despite the effect of CXCR4 blockage in induced as compared with noninduced adrenal glands, we observed no obvious differences in CXCL12-GFP+ (CAR) cell numbers or morphology between groups. Taken together, our results show that the adrenal stroma shares immunophenotypic features with the BM stroma and contains CAR-like CXCL12+ cells. Furthermore, our data are compatible with hematopoietic cells being actively retained in the adrenal niche by CXCL12–CXCR4 signaling.
The murine adrenal gland contains CXCL12+ cells with reticular morphology 3-dimensional (3D) microscopy of murine adrenal glands. Representative 3D sections and optical slices of immunostained adrenal glands from control-treated (A; n = 3) and EMH-induced (B; n = 3) Cxcl12GFP transgenic mice showing endomucin (EMCN; cyan), CXCL12-GFP (yellow), and 4’,6-diamidino-2-phenylindole (DAPI; blue). Scale bars represent 200 μm (A) and 500 μm (B) in the left images, and 40 μm for enlarged sections and optical slices on the right.
The murine adrenal gland contains CXCL12+ cells with reticular morphology 3-dimensional (3D) microscopy of murine adrenal glands. Representative 3D sections and optical slices of immunostained adrenal glands from control-treated (A; n = 3) and EMH-induced (B; n = 3) Cxcl12GFP transgenic mice showing endomucin (EMCN; cyan), CXCL12-GFP (yellow), and 4’,6-diamidino-2-phenylindole (DAPI; blue). Scale bars represent 200 μm (A) and 500 μm (B) in the left images, and 40 μm for enlarged sections and optical slices on the right.
The adrenal stroma is modified by the EMH-induction cocktail
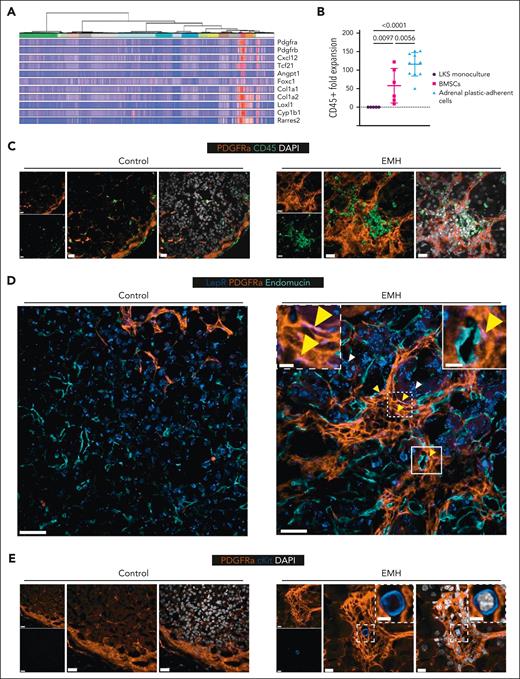
Our data granted further examination of the stroma of the adrenal gland. For this, we examined a recently published publicly available single-nucleus transcriptomics data set of the murine adrenal gland, including 2 adrenal stroma clusters.19 We found that the adrenal stroma expresses Pdgfra (which encodes for the protein PDGFRα) as well as hematopoietic-supportive genes such as Cxcl12 (Figure 5A).
EMH-induced adrenal glands have a modified stromal architecture with PDGFRα clusters hosting hematopoietic foci with rare c-Kit+ HSPCs. (A) Selection of genes enriched in the PDGFRα-expressing cells of the murine adrenal gland (PDGFRα-expressing clusters: mC6 “mesenchymal” in red and mC13 “capsule” in purple, as defined by Bedoya-Reina et al19). (B) Adrenal plastic-adherent cells are supportive of CD45+ cell expansion. Expansion of total hematopoietic cells (CD45+) upon coculture for 7 days of fluorescence-activated cell sorted murine HSPCs (LKS) with a confluent feeder layer of BM stromal cells (BMSCs) obtained from flushed, collagenase-digested bones or adrenal gland, in the absence of additional cytokines. Data are represented as mean ± SD and groups were compared with 1-way ANOVA followed by Holms-Šidák multiple correction test (n = 5 for LKS monoculture, 6 for BMSCs, and 11 for adrenal plastic-adherent cells. Data were obtained in 2 independent experiments). (C) Representative images of control and EMH–induced adrenal glands (n = 2-3 mice per group) stained for CD45 (green), PDGFRα (orange) and DAPI (white). Scale bars represent 20 μm in all instances in panel C. (D) Representative images of control and EMH-induced adrenal glands (n = 2, multiple nodules per adrenal) stained for LEPR (blue), PDGFRα (orange), and endomucin (teal). Yellow arrowheads indicate examples of colocalization of PDGFRα and LEPR (colocalization in pink), with the latter having an elongated pattern marking cells in pericyte position, whereas white arrowheads indicate adrenal parenchyma cells that display a different pattern of LEPR, mostly perinuclear. Scale bars represent 20 μm in all instances in panel D, except for the inset, in which it represents 5 μm. (E) Representative images of control and EMH-induced adrenal glands (n = 3, multiple nodules per adrenal) stained for PDGFRα (orange), cKit (blue), and DAPI (white). Scale bars represent 20 μm in the controls, and 10 μm in the EMH-induced mice. The scale bar in the inset represents 5 μm.
EMH-induced adrenal glands have a modified stromal architecture with PDGFRα clusters hosting hematopoietic foci with rare c-Kit+ HSPCs. (A) Selection of genes enriched in the PDGFRα-expressing cells of the murine adrenal gland (PDGFRα-expressing clusters: mC6 “mesenchymal” in red and mC13 “capsule” in purple, as defined by Bedoya-Reina et al19). (B) Adrenal plastic-adherent cells are supportive of CD45+ cell expansion. Expansion of total hematopoietic cells (CD45+) upon coculture for 7 days of fluorescence-activated cell sorted murine HSPCs (LKS) with a confluent feeder layer of BM stromal cells (BMSCs) obtained from flushed, collagenase-digested bones or adrenal gland, in the absence of additional cytokines. Data are represented as mean ± SD and groups were compared with 1-way ANOVA followed by Holms-Šidák multiple correction test (n = 5 for LKS monoculture, 6 for BMSCs, and 11 for adrenal plastic-adherent cells. Data were obtained in 2 independent experiments). (C) Representative images of control and EMH–induced adrenal glands (n = 2-3 mice per group) stained for CD45 (green), PDGFRα (orange) and DAPI (white). Scale bars represent 20 μm in all instances in panel C. (D) Representative images of control and EMH-induced adrenal glands (n = 2, multiple nodules per adrenal) stained for LEPR (blue), PDGFRα (orange), and endomucin (teal). Yellow arrowheads indicate examples of colocalization of PDGFRα and LEPR (colocalization in pink), with the latter having an elongated pattern marking cells in pericyte position, whereas white arrowheads indicate adrenal parenchyma cells that display a different pattern of LEPR, mostly perinuclear. Scale bars represent 20 μm in all instances in panel D, except for the inset, in which it represents 5 μm. (E) Representative images of control and EMH-induced adrenal glands (n = 3, multiple nodules per adrenal) stained for PDGFRα (orange), cKit (blue), and DAPI (white). Scale bars represent 20 μm in the controls, and 10 μm in the EMH-induced mice. The scale bar in the inset represents 5 μm.
The transcriptomics data prompted us to functionally interrogate the intrinsic hematopoietic-supportive capacity of the uninduced adrenal stroma. For this, we conducted in vitro coculture studies with fluorescence-activated cell–sorted BM-derived HSPCs (LKS cells) which were plated on adrenal stroma obtained by plastic-adherence, then measured the CD45+ cell output after 7 days of coculture in the absence of additional cytokines. LKS cells plated on adrenal plastic-adherent cells produced a larger CD45+ progeny than those plated on monolayers of BM stromal cells or cultured alone (Figure 5B). Our results suggest that plastic-adherent cells from the adrenal stroma may be intrinsically supportive of hematopoiesis.
Then, we examined in situ EMH-induced adrenal glands for stromal markers known to be associated with the hematopoietic BM niche, including PDGFRα, in line with previous reports of mesoderm-derived stromal cells regulating the emergence of the AGM hematopoietic niche.20 Indeed, staining for PDGFRα showed a marked reorganization of the adrenal cortical stroma in the EMH-induced organs surrounding foci of small, tightly packed nuclei that were morphologically reminiscent of EMH in hematoxylin and eosin–stained sections (supplemental Figure 3A). The cell clusters within the PDGFRα stromal nodules were positive for CD45+, confirming the identity of the hematopoietic foci (Figure 5C). Moreover, and surprisingly, we observed a certain degree of colocalization of PDGFRα with leptin receptor (LEPR) exclusively in the stromal clusters from EMH-induced adrenal glands, with PDGFRα+LEPR+ stromal cells located in pericyte position as defined by endomucin-positive endothelial cells (Figure 5D, inset; single-channel images and the DAPI [4’,6-diamidino-2-phenylindole] overlay are provided in supplemental Figure 3B). Because LEPR is a commonly used marker to identify the BM stroma,21 our results hint at the possibility of inducing a BM stroma–like phenotype in the adrenal stroma. Finally, we interrogated the EMH-induced adrenal glands for cells positive for markers of HSPCs in the PDGFRα nodules. For this, we stained sections with c-Kit and PDGFRα and observed that the nodules did contain rare c-Kit+ cells, corroborating our transplantation data (Figure 5E).
Together, our data show that the EMH induction cocktail triggers marked changes in the adrenal stroma architecture, with formation of PDGFRα+LEPR+/− clusters encompassing hematopoietic foci capable of hosting rare c-Kit+ HSPCs, and thus potentially enhancing the intrinsic hematopoietic-supportive capacity of the adrenal stroma in vivo.
Human myelolipoma is positive for BM stroma markers and contains CXCL12+ reticular cells
Myelolipoma, a benign tumor composed of adipose and hematopoietic tissues, is frequently found in the adrenal gland, particularly in the context of endocrine disorders that associate elevated ACTH levels. We hypothesized that human myelolipomas might recapitulate a phenomenon like the one we observe in the adrenal gland of mice treated with our EMH-induction cocktail. For this, we retrieved myelolipoma samples originally collected at the Centre Hospitalier Universitaire Vaudois, Lausanne, Switzerland. The clinical characteristics of our myelolipoma cohort are summarized in supplemental Table 1, including the anatomical origin of the myelolipomas, of which 60% were adrenal and 40% were pelvic or retroperitoneal. We then compared our samples with the published registry, gathering all reported data cases of adrenal myelolipoma, recently published by Decmann et al22 (Table 1). Surprisingly, we found that 20% of the patients in our cohort had a history of splenectomy (n = 2/10) whereas the previously published prevalence of splenectomy in the general population is ∼0.4%.23
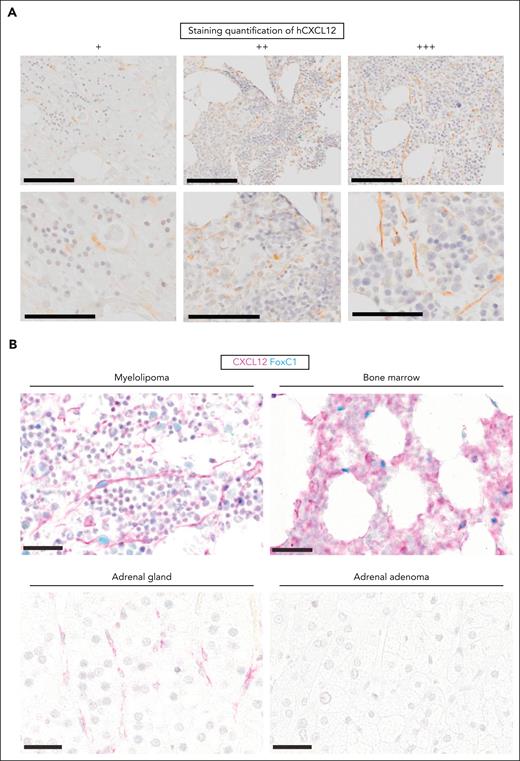
We performed IHC studies of our myelolipoma samples using a panel of markers designed to evaluate known components of the human BM stroma, namely CD73, CD90, CD146, CD271, CXCL12, and Nestin, as well as CD34 to target hematopoietic progenitors.24-26
Using paraffin-embedded samples stored from the diagnostic workup of our patients, we stained consecutive sections and examined the presence of positive cells in IHC. We included, as controls, human BM and an adrenal adenoma. CD34 and CD90 were expressed in only a fraction of our samples. CD73, CD146, CD271, and Nestin were expressed across all samples (supplemental Figures 4 and 5A). Notably, CXCL12 was present in all myelolipoma samples (Figure 6A ; Table 1), but not in the adrenal adenoma controls (n = 3). Furthermore, CXCL12+ cells were of reticular morphology and coexpressed the transcription factor FOXC1, the best characterized master regulator of hematopoietic support factor expression in BM niche cells,27 at least to a similar extent as BM stroma, which was not the case for healthy adrenal glands or adrenal adenomas (Figure 6B). This stain was specific, as illustrated in supplemental Figure 5B-C. Taken together, our results indicate the reproducible detection of CXCL12+ FOXC1+ stromal cells in human myelolipoma samples with similar characteristics to BM stroma. Thus, our data support the use of adrenal myelolipomas as a surrogate to understand the composition of an inducible supportive hematopoietic niche, which mirrors our inducible adrenal niche model.
CXCL12+ cells of stromal morphology are present in human myelolipoma and coexpress FOXC1. (A) Representative images of CXCL12 standard IHC staining (brown) of human myelolipoma samples corresponding, from left to right, to patients I, C, and D (see Table 1 for details). Scale bars correspond to 200 μm and 100 μm in the top and bottom panels, respectively. (B) Double chromogenic immunohistochemical stain for CXCL12 (pink) and FOXC1 (light blue) in human myelolipoma, BM, healthy adrenal gland, and adrenal adenoma. Scale bar represents 30 μm at all instances.
CXCL12+ cells of stromal morphology are present in human myelolipoma and coexpress FOXC1. (A) Representative images of CXCL12 standard IHC staining (brown) of human myelolipoma samples corresponding, from left to right, to patients I, C, and D (see Table 1 for details). Scale bars correspond to 200 μm and 100 μm in the top and bottom panels, respectively. (B) Double chromogenic immunohistochemical stain for CXCL12 (pink) and FOXC1 (light blue) in human myelolipoma, BM, healthy adrenal gland, and adrenal adenoma. Scale bar represents 30 μm at all instances.
Discussion
Here, we show that the adrenal gland can be transformed into a hematopoietic-supportive environment and used as a model to study the minimal stromal components of a non-ossified de novo hematopoietic niche. The EMH-induced adrenal niche contained CXCL12+ cells with classical reticular morphology, concomitant to the formation of PDGFRα+LEPR+/− stromal clusters, which associated to tightly packed, cobblestone-like hematopoietic colonies containing rare c-Kit+ HSPCs, including serially transplantable stem cells. Our findings are supported by the presence of CAR-like CXCL12+FOXC1+ reticular cells in human adrenal myelolipoma, associated to CD34+ HSPCs.
The observation that the adult adrenal gland can support hematopoiesis upon hormonal stimulation is particularly interesting because the adrenal cortex originates from the AGM, the structure that gives rise to definitive HSCs during embryonic development, with critical involvement of environmental cues provided by mesoderm-derived PDGFRα+ stromal cells.20 The adrenal gland is therefore ontogenically related to a hematopoietic-supportive structure and can be transformed into an adult niche, a phenomenon that has been clearly described in the spleen and the liver.2 Moreso, it has been recently shown in a human cell atlas of fetal gene expression, that the fetal adrenal gland contains limited erythropoiesis, pointing to a previously overlooked hematopoietic-supportive capacity of the adrenal gland.28 Furthermore, the propensity of myelolipoma to develop in the adrenal gland suggests a specific hematopoietic-supportive population in this location. Adrenal myelolipoma has been proposed to originate from a mesenchymal progenitor cell giving rise to the stromal compartment and then recruiting hematopoietic cells,29 something that would be in line with our observations.
We initially attempted to fully recapitulate myelolipomas in mice with both the adipocytic compartment and hematopoietic cells. Based on historical publications using rats as models and crude pituitary extracts as stimulants, we developed a model using chemically defined hormonal stimulation in splenectomized mice. G-CSF was used to simulate stress hematopoiesis. G-CSF treatment alone directly stimulates HSPCs proliferation and mobilization, accelerating exit of severe neutropenia by an average of 3 to 6 days in humans.30,31 In accordance with the original description of Selye and Stone,9 our cocktail also contained testosterone. The effect of androgens as a stimulant of hematopoiesis has been thoroughly described and is used in patients to treat BM insufficiency, specifically in the context of telomeropathies and Fanconi anemia.32-35 Instead of pituitary extracts, ACTH daily was chosen to induce EMH in the adrenal gland because the incidence of myelolipoma increases several-fold in patients suffering from congenital adrenal hyperplasia, a disease of the cortisol axis that increases the levels of ACTH.22 Consequently, our induction cocktail is based on stimulation of both the adrenal gland, with ACTH, and the hematopoietic system, with G-CSF and testosterone. We, however, failed to observe adipocytes in our model, which suggests that mature adipocytes might not be necessary for hematopoietic support in the adrenal EMH. Finally, we found that splenectomy was necessary for the full induction of EMH in the adrenal gland. As the spleen is a known site of physiological EMH in mice,36 we speculate that its presence might retain circulating HSPCs that would otherwise colonize the adrenal gland in our model.
In our collection of human adrenal myelolipoma samples, patients presented similar characteristics as published in the literature. Interestingly, we found an overrepresentation of patients with splenectomy in our myelolipoma cases. Although the cohort is small, and this could be an incidental finding, it could play a role in the development of myelolipoma. An in-house IHC-based panel, based on markers described for the BM stroma, consistently showed the presence of CXCL12-expressing cells with reticular morphology, which coexpressed FOXC1 in the myelolipomas. Both proteins have been reported as markers of BM stromal cell populations capable of providing hematopoietic support, with CXCL12 having a known mechanistic role in this function, and FOXC1 acting as a master regulator of hematopoietic-supporting stromal niche cells.27
In conclusion, we present our model as a novel tool to increase our understanding of the physiology of hematopoietic support and to facilitate the study of inducible, adult-specific niche models. From a phylogenetic standpoint, examples of adult-specific hematopoietic niches outside of the BM exist in vertebrate evolution, and appear as early as in jawless fish in the form of the dorsal fat body.37 The composition of what constitutes the simplest unit of hematopoietic niche, supporting both HSC self-renewal and progenitor expansion, remains largely unknown. Because the exact composition needed to recapitulate enough complexity of the hematopoietic microenvironment for it to be functional is still undefined, further understanding of minimalistic niches, like the inducible boneless adrenal niche we report, has the potential to aid in the development of biomedical and tissue engineering applications.
Acknowledgments
The authors thank Josefine Tratwal, Paolo Bianco, Mukul Girotra, Shanti Rojas-Sutterlin, Marian Manongdo, and Markus Manz for help in the inception of the project, and for providing critical advice along its development. This project would not have been possible without the support of the École Polytechnique Fédérale de Lausanne (EPFL) animal facility (Centre of PhenoGenomics) and the animal caretakers, in particular, Laetitia Cagna, Margaux Mouchet, and Pierre Dodane. The authors thank the EPFL and UNIL flow cytometry facilities, in particular Danny Labes and the members of the EPFL histology facility, and the EPFL Biomolecular Screening Facility. The authors thank Nathalie Piazzon (Institut de Pathologie Biobank, University of Lausanne [UNIL]/Centre Hospitalier Universitaire Vaudois, Lausanne) for facilitating the access to human myelolipoma samples. The authors thank Takashi Nagasawa, Graduate School of Frontier Biosciences and Graduate School of Medicine, Osaka University, Japan, for the generous gift of the CXCL12/GFP knockin mice.
F.S. was funded by the Swiss National Science Foundation (SNSF) MD-PhD grant 183986. O.N. was funded by SNSF Professorship grants PP00P3_144857, PP00P3_176990, and PP00P3_183725 and, together with A.A.-C., by UNIL. C.N.-A. was funded by the Consolidator Grant from the European Research Council (ERC-Co2019 86580). This research was funded, in whole or in part, by the Swiss National Science Foundation (grant numbers: 183986, PP00P3_144857, PP00P3_176990, and PP00P3_183725).
Authorship
Contribution: O.N. and F.S. conceived the ideas and obtained funding for the project; F.S., with the help of A.A.-C., S.F.L., and A.O., performed all the experiments. F.S., O.N., and A.A.-C. analyzed the results and wrote the manuscript; A.V. and C.N.-A. performed and analyzed all whole-mount confocal microscopy imaging presented in this manuscript; R.S., C.B., J.S.-D., and L.d.L. set up the immunostaining panel for human myelolipomas; S.G. assessed the myelolipoma images; F.S. and O.N. compiled the clinical data; F.S. developed the experimental pipelines and executed the in vivo experiments; and A.A.-C. was involved in tissue analysis, data analysis, and finalization of the project.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Olaia M. Naveiras, Department of Biomedical Sciences, University of Lausanne, Bugnon 27, 1011 Lausanne, Switzerland; email: olaia.naveiras@unil.ch.
References
Author notes
F.S. and A.A.-C. are joint first authors and contributed equally to this study.
Data sets have been deposited at https://zenodo.org/records/11273437.
The online version of this article contains a data supplement.
There is a Blood Commentary on this article in this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal