Visual Abstract
Hereditary hemorrhagic telangiectasia (HHT; Osler-Weber-Rendu disease) affects 1 in 5000 persons, making it the second most common inherited bleeding disorder worldwide. Telangiectatic bleeding, primarily causing recurrent epistaxis and chronic gastrointestinal bleeding, is the most common and most important manifestation of this multisystem vascular disorder. HHT-associated bleeding results in substantial psychosocial morbidity and iron deficiency anemia that may be severe. Although there remain no regulatory agency–approved therapies for HHT, multiple large studies, including randomized controlled trials, have demonstrated the safety and efficacy of antifibrinolytics for mild-to-moderate bleeding manifestations and systemic antiangiogenic drugs including pomalidomide and bevacizumab for moderate-to-severe bleeding. This has led to a recent paradigm shift away from repetitive temporizing procedural management toward effective systemic medical therapeutics to treat bleeding in HHT. In this article, 4 patient cases are used to illustrate the most common and most challenging presentations of HHT-associated bleeding that hematologists are likely to encounter in daily practice. Built on a framework of published data and supported by extensive clinical experience, guidance is given for modern evidence–based approaches to antifibrinolytic therapy, antiangiogenic therapy, and iron deficiency anemia management across the HHT disease severity spectrum.
Introduction
With a prevalence of 1 in 5000 people, hereditary hemorrhagic telangiectasia (HHT; Osler-Weber-Rendu disease) is the second most common inherited bleeding disorder worldwide. Resulting from mutations in the activin receptor-like kinase 1 (ALK1 or ACVRL1) signaling pathway, HHT is an autosomal dominant disease of angiogenic dysregulation, manifesting clinically as bleeding mucocutaneous telangiectasias and visceral arteriovenous malformations (AVMs).1 Mucocutaneous telangiectasias result in recurrent, often severe epistaxis as well as chronic gastrointestinal bleeding, both of which frequently result in iron deficiency anemia that may be iron infusion and/or red cell transfusion dependent.2 Visceral AVMs can develop in almost any organ but most commonly occur in the liver (70%), lung (50%), and brain (10%-20%). These lesions may result in varied morbid or fatal complications including intracerebral hemorrhage, embolic stroke, hemoptysis, high-output heart failure, chronic liver disease, and others.
Because bleeding is nearly universal in HHT and can be severe, livelihood-threatening, or life-threatening, patients rank bleeding as the most important complication of their disease by a wide margin (with AVMs second and anemia third).3 So, although comprehensive HHT care requires a multidisciplinary team analogous to comprehensive hemophilia care,4 hematologists, hematologist-oncologists, and internists with expertise in antiangiogenic, antifibrinolytic, and IV iron treatments generally serve as the medical home for these patients given their ongoing needs for bleeding and anemia management (this role can also be fulfilled by other specialists who explicitly focus their clinical practice on HHT in certain centers). Recurrent epistaxis, which ultimately affects nearly all patients with HHT, may alone result in major negative impact in health-related quality of life (HRQoL), precipitating anxiety and mood disorders, posttraumatic stress disorder, social isolation and social phobia, and unemployment.5,6 Although in previous decades the treatment paradigm for both epistaxis and gastrointestinal bleeding centered around procedural management, a paradigm shift to treatment of bleeding with systemic therapy has occurred.7,8 This shift has been spurred by a vastly improved understanding of physiologic angiogenesis9,10 and HHT disease pathophysiology11 in the past few decades and the availability of systemic antiangiogenic therapies originally developed to treat malignancies that have disease-modifying properties in HHT. These therapies are successfully being repurposed to treat this neglected bleeding disorder.12
Given that HHT remains without any US Food and Drug Administration– or European Medicines Agency–approved therapies,12 however, and that the use of systemic therapies to treat HHT remains relatively novel and entirely off-label, guidance for the use of these agents is essential for their safe and proper clinical application. Therefore, using a series of typical clinical presentations, this article provides guidance for the treatment of bleeding in adults with HHT, incorporating the best available evidence, including data from recently published randomized clinical trials, along with extensive clinical experience with the use of the discussed agents in several hundred patients with HHT. The systemic therapies with robust evidence for effectiveness and safety, including bevacizumab, pomalidomide, and antifibrinolytic agents will be the focus, with some discussion of agents with more limited evidence (including thalidomide, pazopanib, and somatostatin analogs).
HHT-associated bleeding: severity assessment and management goals
Bleeding severity assessment
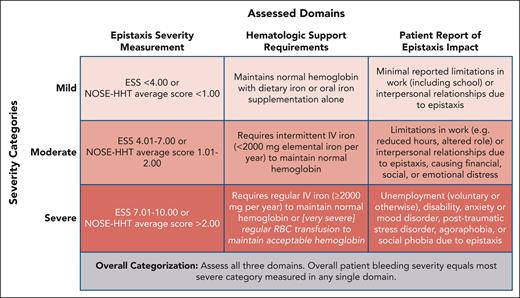
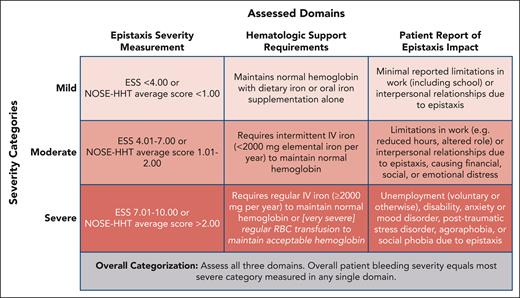
Bleeding in HHT is heterogeneous, with substantial variation from patient to patient.2,6 It is also typically progressive, worsening with advancing age.13 Therefore, when categorizing the severity of a patient’s HHT-associated bleeding, 3 domains should be assessed (Figure 1) at initial evaluation with reassessment at least once yearly. This method uses epistaxis severity measurement with a validated clinical instrument plus the patient report of epistaxis impact and considers total quantity of blood loss through the nose and gastrointestinal tract via assessment of hematologic support requirements. No studies have yet been published rigorously subclassifying bleeding severity in HHT. In my experience directing a US HHT Center of Excellence, however, according to the categorization in Figure 1, at any given point in time ∼40% of patients have mild bleeding, 40% have moderate bleeding, and 20% have severe bleeding. Within the severe bleeding category, an important minority—perhaps 5% of all patients—have very severe bleeding, defined as a requirement for regular red cell transfusion to maintain an acceptable hemoglobin. Patients with mild-to-moderate bleeding are usually successfully managed with local noninvasive measures and antifibrinolytic therapies, and those with moderate-to-severe bleeding frequently require disease-modifying antiangiogenic therapies for satisfactory bleeding management.8
Assessment of bleeding severity in HHT. All 3 domains should be assessed at initial evaluation and assessment should be repeated at least once yearly. RBC, red blood cell.
Assessment of bleeding severity in HHT. All 3 domains should be assessed at initial evaluation and assessment should be repeated at least once yearly. RBC, red blood cell.
Epistaxis: assessment and goals of management
The primary goal of epistaxis management is reduction in frequency, intensity, and duration of epistaxis such that its impact on the patient’s employment, social and personal life, and overall HRQoL is minimal. Occasionally, near or complete elimination of epistaxis is possible. I assess epistaxis severity using 1 or both of 2 validated HHT-specific epistaxis severity instruments (only 1 is necessary). The epistaxis severity score (ESS) is a 6-question instrument scored between 0.00 (no epistaxis) and 10.00 (the worst possible epistaxis) that may be assessed rapidly in the clinic, although it uses complex arithmetic to score and is best calculated using a free online calculator (such as the calculator hosted at https://curehht.org/resource/epistaxis-severity-score/).14 The minimal clinically important difference (MCID) for the ESS is 0.71 points.15 The ESS is assessed over a specific time period, usually the past 1 to 3 months. The Nasal Outcome Score for Epistaxis in HHT (NOSE-HHT) is a validated epistaxis-specific instrument with 29 items each scored from 0.00 (no impact) to 4.00 (worst possible impact) assessing epistaxis severity over the past 2 weeks and is scored by averaging the score of all answered questions.16 The MCID for NOSE-HHT is 0.46 points. A free online calculator for NOSE-HHT is available (https://outcomesresearch.github.io/nose-hht/). Given the ease and rapidity of the ESS, it is simple to incorporate into routine practice and calculate at each visit to monitor epistaxis severity over time. Given its length, routine clinical use of NOSE-HHT is best accomplished by administration of the instrument on paper or electronic tablet while the patient awaits the visit with the clinician.
Gastrointestinal bleeding: assessment and goals of management
The primary goal of gastrointestinal bleeding management is to eliminate or minimize anemia, its impact on health and HRQoL, and the need for chronic hematologic support to manage it. Chronic gastrointestinal bleeding should be suspected in any patient with anemia and hematologic support requirements out of proportion to epistaxis severity. Comprehensive endoscopic evaluation (including esophagogastroduodenoscopy, colonoscopy, and video capsule endoscopy or enteroscopy) can usually establish the presence or absence of gastrointestinal telangiectasias, which may be present only in jejunum or ileum beyond the reach of a typical upper endoscopy. The presence of these lesions, with or without active bleeding at the time of endoscopic visualization, in combination with anemia out of proportion to epistaxis severity is adequate for a diagnosis of chronic HHT-associated gastrointestinal bleeding.8
Iron deficiency and iron deficiency anemia: assessment and goals of management
Regular monitoring of hemoglobin, red cell parameters, and iron status, with aggressive management of iron deficiency and iron deficiency anemia, are an essential cornerstone of HHT management. Approximately half of patients with HHT are anemic at any given time and most ultimately develop iron deficiency anemia recurrently over the course of life.2,8 Women with HHT must contend with both HHT-associated epistaxis and gastrointestinal bleeding as well as menstrual bleeding, and women with HHT appear to have a higher incidence of heavy menstrual bleeding compared with the general population17,18 (although perhaps not as high as women with von Willebrand disease19). Presently, management of heavy menstrual bleeding in women with HHT mirrors that of the general population, although the true incidence of uterine AVMs and endometrial telangiectasias and their contribution to bleeding is currently unknown and remains an area of active research. The interval for monitoring with hemoglobin, serum ferritin, and transferrin saturation in patients with HHT should be individualized and can range between once to twice weekly for red cell transfusion–dependent patients, to once yearly for those with no history of anemia8; in my experience, on average, patients with HHT require this monitoring every 3 to 4 months. Pregnant patients (and those planning pregnancy) with moderate or severe HHT-associated bleeding require very close monitoring, typically monthly throughout pregnancy, and usually require aggressive management with IV iron to maintain an iron replete status given the known adverse impact of maternal iron deficiency on fetal neurologic development.20
Acceptance of chronic iron deficiency or iron deficiency anemia is inappropriate in patients with HHT. There are some patients with very severe, rapid bleeding who cannot maintain a normal hemoglobin despite aggressive iron infusion (1000-2000 mg infused elemental iron per month) or those who require red cell transfusion to maintain an acceptable hemoglobin7,8; barring absolute contraindications, systemic antiangiogenic therapy is indicated in this group of patients to reduce bleeding and allow for resolution of anemia (it is also indicated in other patients with moderate-to-severe bleeding, as described below). The definition of “acceptable hemoglobin” in these very severely bleeding patients is individualized (usually between 8-10 g/dL), and higher thresholds for transfusion are required in HHT compared with the general population, given that bleeding is chronic and persistent. In patients without rapid bleeding, IV iron infusions function as delayed-onset red cell transfusions because patients with HHT have normal bone marrow function in the absence of comorbid marrow defect.21 To avoid iron deficiency and its complications, I target a serum ferritin of ≥50 ng/mL and transferrin saturation of ≥20% in all such patients with HHT. Table 1 lists typical IV iron formulations and considerations for their use in HHT. In my experience, chronic borderline iron deficiency (persistent measurements below these targets without anemia) with iron deficiency symptoms is common in patients with HHT maintained on oral iron alone and should not be accepted when IV iron is safe and effective. In addition to impairing HRQoL, chronic iron deficiency has been associated with increased thromboembolic risk in patients with and without HHT.22-25
Management of mild-to-moderate epistaxis
Patient 1 (mild-to-moderate epistaxis)
A 38-year-old paralegal with HHT presents for evaluation. Recurrent epistaxis has required her to adopt an all-dark wardrobe and leave work early 3 times in the past 6 months. She additionally describes several nosebleeds during romantic and social outings since her last visit, causing embarrassment and isolation. Her ferritin was measured at 16 ng/mL 6 months ago, prompting initiation of ferrous sulfate 325 mg (65 mg elemental iron) tablets, which she takes once daily. She currently denies symptoms of anemia or iron deficiency. Her hemoglobin is 13.9 g/dL, serum ferritin is 54 ng/mL, and transferrin saturation is 24%. Her 1-month ESS is 4.35, and her NOSE-HHT average score is 0.92, despite good adherence to a daily nasal moisturization regimen.
Nasal moisturization and local nasal medical and surgical therapies
All patients with HHT-associated epistaxis should adopt a daily nasal moisturization regimen including topical application of nasal saline solution or gels and air humidification (particularly in sleeping environments).8 This alone can modestly improve epistaxis and should be attempted before initiation of therapy in patients with mild epistaxis.29 Although local ablative and procedural therapies (such as laser therapies, electrosurgery, radiofrequency ablation, sclerotherapy, and others) usually provide partial and temporary improvement in epistaxis,8 the tissue injury they necessarily cause may stimulate increased local vascular endothelial growth factor (VEGF)–driven telangiectasia formation in the nasal mucosa and ultimately recurrence of bleeding more severe than before the procedure.30 Recent clinical studies have substantiated these concerns.31 Therefore, the modern HHT treatment paradigm has departed from cyclic repetition of local hemostatic procedures. Topical drug therapies directly to the nasal mucosa (such as tranexamic acid, estrogen, and bevacizumab nasal sprays; tacrolimus ointment; or submucosal bevacizumab injections) have all failed to achieve their primary end points against placebo in randomized clinical trials.29,32-34 Therefore, I use systemic medical therapy to treat chronic recurrent epistaxis and reserve local ablative/surgical therapies for either (1) acute severe bleeding requiring immediate hemostasis, or (2) those patients requiring antithrombotic therapy (as described in the case of patient 4 below).
Oral antifibrinolytic therapy
Oral antifibrinolytic therapy, particularly tranexamic acid, has robust evidence for efficacy in the treatment of mild-to-moderate epistaxis in HHT, and would be the optimal initial therapeutic choice for patient 1. Two randomized, placebo-controlled clinical trials enrolling a total of 140 patients with HHT and overall moderate epistaxis found modest but significant improvements in epistaxis duration and intensity with routine oral tranexamic acid use,35,36 and a retrospective cohort study described modest effectiveness of oral ε-aminocaproic acid and tranexamic acid in reducing HHT-associated gastrointestinal bleeding.37 Antifibrinolytic therapy alone is rarely adequate in patients with severe epistaxis or more than mild gastrointestinal bleeding, and therefore I offer it primarily to patients with mild-to-moderate epistaxis as an initial systemic therapy. The major drawback of oral antifibrinolytic therapy is dosing frequency and pill burden, because it must be dosed 3 to 4 times daily (eg, 1300 mg tranexamic acid every 8 hours or 1000 mg ε-aminocaproic acid every 6 hours) given its short half-life in order to maintain a consistent effect. Common adverse events include nausea and diarrhea, which are usually mild and temporary. Antifibrinolytic therapy carries a theoretical thromboembolic risk, on which patients should be counseled before initiation. However, randomized clinical trials and observational studies in HHT have not reported any evidence suggesting increased thromboembolic risk in patients treated with doses in the range of 3 to 4 g per day for extended durations.35-38
Because antiangiogenic therapy carries additional potential risks in growing children, oral antifibrinolytics are the primary systemic bleeding therapy currently prescribed to children with HHT. Because bleeding in HHT is progressive and generally worsens with age, the vast majority of pediatric patients with bleeding have mild-to-moderate epistaxis and are successfully managed with this approach; in cases in which it fails, local ablative procedures can be considered.
Management of moderate-to-severe epistaxis and gastrointestinal bleeding
Patient 2 (moderate-to-severe epistaxis)
A 46-year-old chef with HHT presents for evaluation. He recently went on leave from his job because of epistaxis. He describes multiple gushing nosebleeds daily, which routinely ruin plates of food in his kitchen and have caused him to develop considerable anxiety around having nosebleeds at work. His 1-month ESS today is 6.16, and his NOSE-HHT average score is 3.10. He has no known gastrointestinal bleeding. On laboratory evaluation today, his hemoglobin is 14.5 g/dL, serum ferritin is 81 ng/mL, and transferrin saturation is 26%. He is receiving ferumoxytol 510 mg every 4 months to treat and prevent recurrence of iron deficiency anemia. He has undergone multiple local ablative procedures previously to treat epistaxis in prior years, which he described as painful and temporizing with bleeding recurring after 2 to 3 months at similar or greater intensity than before the procedure. He previously tried oral tranexamic acid with minimal improvement.
Selection of systemic antiangiogenic therapy in HHT
Because increased VEGF drives telangiectasia and AVM formation in HHT mouse models and normalization of VEGF suppresses their formation,39,40 antiangiogenic therapy is believed to target the underlying pathogenesis of telangiectasias and AVMs in HHT. Certain antiangiogenic therapies have become established therapeutic modalities in HHT, and others under active investigation are considered quite promising. Antiangiogenic therapy is often referred to as “disease-modifying” therapy in HHT because unlike other treatments, successful antiangiogenic therapy may induce involution of mucocutaneous telangiectasias (which can often be clearly visualized on the skin, oral cavity, and nasal and gastrointestinal mucosa)41 and may potentially arrest progression or even induce regression of certain visceral AVMs (primarily hepatic AVMs).42-44 Theoretically, antiangiogenic therapy may also prevent formation of additional telangiectasias or AVMs that would have formed in its absence, although this has not been proven. Numerous systemic antiangiogenic agents have been developed and licensed for the treatment of malignancies, but only a small portion of these have been evaluated in HHT.7 When disease-modifying therapy is indicated, whenever they are available, clinical trials of novel or repurposed disease-modifying agents should always be offered to patients first. Two agents, bevacizumab and pomalidomide, have robust evidence from large studies demonstrating safety and efficacy in HHT45,46 (Table 2) and are the primary agents I use off-label to treat moderate-to-severe HHT-associated bleeding in patients not obtaining treatment on a clinical trial of another agent. An alternative agent, thalidomide, has been evaluated in smaller studies in patients with HHT with demonstrated efficacy47-50 (as well as larger studies in patients with bleeding intestinal angiodysplasia who do not have HHT51) and can be considered in place of pomalidomide in which the latter is unavailable. Thalidomide, unlike pomalidomide, carries a substantial risk of irreversible peripheral neuropathy (a risk that increases with increasing duration of use).52,53 Thalidomide also has a higher thromboembolic risk than pomalidomide.54 These are both major concerns in the setting of HHT, for which indefinite treatment is required to maintain hemostasis, and why I do not use thalidomide routinely to treat bleeding in HHT. Additional agents are currently under investigation (Table 2).
Both pomalidomide and bevacizumab may be effective in treatment of epistaxis and gastrointestinal bleeding, and if either of these is ineffective in a given patient, the other may be effective and should be considered. It is not currently possible to predict whether an agent will be successful in a given patient before its use. In my own experience treating >100 patients with moderate-to-severe HHT-associated bleeding with these 2 therapies, however, I have found greater success with bevacizumab to treat gastrointestinal bleeding and greater success with pomalidomide to treat epistaxis. Therefore, after considering contraindications (Table 3), I generally favor pomalidomide as the first-line antiangiogenic when epistaxis is the greater problem (as in patient 2) and bevacizumab when gastrointestinal bleeding is the greater problem (as in patient 3 below), recognizing that both bleeding sources often coexist in patients with moderate-to-severe HHT-associated bleeding. If pomalidomide is not available for a patient in whom epistaxis is the greater problem, I recommend bevacizumab over thalidomide as a first-line antiangiogenic owing to the limited data and substantially less-favorable toxicity profile of the latter.
All antiangiogenic therapies carry a theoretical thrombotic risk, which should be discussed when consenting the patient for therapy.54 In the published literature, however, thromboembolic rates with use of these agents have been much lower in patients with HHT (Table 2) than patients with cancer, including in patients treated for several years, supporting their routine and extended-duration use.18,54 Additionally, some patients who initially respond to either bevacizumab or pomalidomide may lose this response for unknown reasons, usually after ≥2 years of therapy. In this circumstance, another disease-modifying agent should be tried, because it may be effective.
Use of pomalidomide to manage HHT-associated bleeding
Pomalidomide is an oral immunomodulatory imide drug with antiangiogenic and anti-inflammatory properties primarily used for the treatment of multiple myeloma. In a large, multicenter National Institutes of Health–sponsored randomized, double-blind, placebo-controlled phase 2 trial of pomalidomide to treat HHT-associated bleeding (PATH-HHT), pomalidomide was found to be safe and efficacious in reducing epistaxis and improving HRQoL in patients with HHT. Although the full manuscript describing the final results of the PATH-HHT study is yet to be published, primary results from this study have been published in abstract form.45 Therefore, data from the published abstract is described in this manuscript. The trial was initially planned to enroll 159 patients randomized 2:1 to pomalidomide or placebo but was closed to enrollment early when a planned interim analysis achieved prespecified early stopping criteria for efficacy (144 patients were enrolled). Patients treated with pomalidomide had a statistically significant improvement in ESS compared with placebo, an improvement that was also highly clinically relevant (a mean of 1.84-point reduction, >2.5 times the MCID for the ESS). A similarly clinically relevant improvement was also observed in an HHT-specific HRQoL instrument. The most common adverse events with pomalidomide (each occurring in ∼30%-50% of patients) included constipation/diarrhea, mild-to-moderate neutropenia, and rash. Venous thromboembolism was rare and occurred in similar proportions of patients in both pomalidomide and placebo groups.
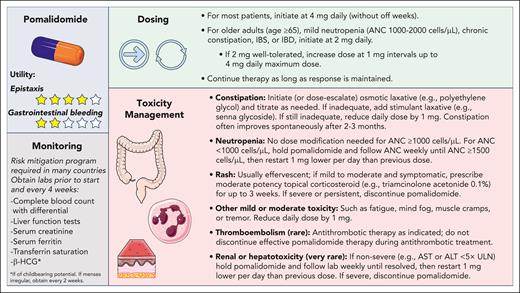
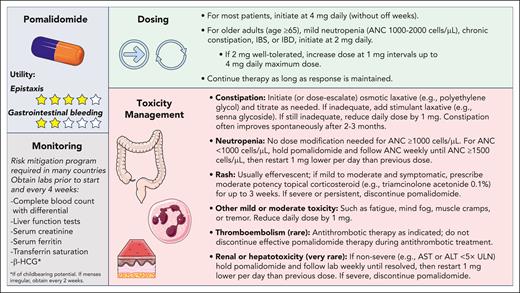
My approach to dosing, monitoring, and toxicity management for pomalidomide in HHT is illustrated in Figure 2. In contrast to typical dosing in multiple myeloma, pomalidomide in HHT is dosed daily without off weeks. In contrast to the PATH-HHT protocol (which initiated all patients at 4 mg daily), I initiate older adults (age of ≥65 years) or those with preexisting chronic gastrointestinal illness (such as chronic constipation, irritable bowel syndrome, or inflammatory bowel disease) or mild neutropenia (absolute neutrophil count between 1000-2000 cells per μL) at 2 mg daily, dose-escalating to up to 4 mg daily as tolerated to maximize hemostatic effect.
Approach to dosing, monitoring, and toxicity management for pomalidomide in HHT. ANC, absolute neutrophil count; ALT, alanine aminotransferase; AST, aspartate aminotransferase; HCG, human chorionic gonadotropin; IBD, inflammatory bowel disease; IBS, irritable bowel syndrome; ULN, upper limit of normal.
Approach to dosing, monitoring, and toxicity management for pomalidomide in HHT. ANC, absolute neutrophil count; ALT, alanine aminotransferase; AST, aspartate aminotransferase; HCG, human chorionic gonadotropin; IBD, inflammatory bowel disease; IBS, irritable bowel syndrome; ULN, upper limit of normal.
Patient 3 (moderate-to-severe gastrointestinal bleeding)
A 63-year-old schoolteacher with HHT presents for evaluation. She has mild epistaxis (ESS, 3.33) but has iron infusion–dependent anemia because of chronic gastrointestinal bleeding. She has undergone 8 endoscopies over the past 5 years, with intermittent argon plasma coagulation and endoscopic clipping of actively bleeding lesions but never with durable anemia resolution. She has required progressively increasing IV iron requirements to maintain a normal hemoglobin. She has received 6000 mg elemental iron over the past year (1000 mg ferric derisomaltose every 2 months). Today, 2 months from her last iron infusion, she is pale and complains of fatigue, mind fog, and a craving for ice. Her laboratory evaluation reveals a hemoglobin of 9.7 g/dL, serum ferritin of 8 ng/mL, and transferrin saturation of 6%.
Use of bevacizumab to manage HHT-associated bleeding
Bevacizumab is an IV administered humanized immunoglobulin G1 monoclonal antibody directed against VEGF-A. In a large 12-center retrospective observational study of systemic bevacizumab to treat HHT-associated bleeding (InHIBIT-Bleed), bevacizumab was observed to be safe and effective in improving hemoglobin and reducing iron infusion and red cell transfusion requirements as well as epistaxis severity in 238 patients with HHT (Table 2).46 The most common adverse events observed with use of systemic bevacizumab in HHT are hypertension (15%-25%), proteinuria (5%-10%), and myalgia/arthralgia (5%-10%). Bevacizumab would be an ideal first-line antiangiogenic therapeutic choice for patient 3.
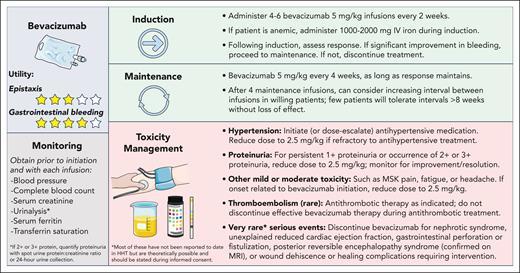
My approach to dosing, monitoring, and toxicity management for bevacizumab in HHT is illustrated in Figure 3. Patients undergo 4 to 6 induction infusions every 2 weeks, during which hemostatic effectiveness and tolerance is assessed. Patients doing well on treatment then proceed to maintenance therapy to preserve the treatment effect. Continuous maintenance (prophylactic therapy, scheduled administration of a bevacizumab infusion every 4-8 weeks) appears to be superior to intermittent maintenance (on-demand therapy, holding therapy until evidence of recurrent iron deficiency, anemia, or worsening bleeding occurs) at maintaining hemoglobin and bleeding control at the cost of greater drug exposure and is my preferred maintenance strategy. Intermittent maintenance, however, is a reasonable alternative and may be preferred by certain patients (for logistical or financial reasons).46
Approach to dosing, monitoring, and toxicity management for bevacizumab in HHT. MSK, musculoskeletal; MRI, magnetic resonance imaging.
Approach to dosing, monitoring, and toxicity management for bevacizumab in HHT. MSK, musculoskeletal; MRI, magnetic resonance imaging.
Special situations and treatment-refractory HHT-associated bleeding
Patient 4 (moderate epistaxis but requiring antithrombotic therapy)
A 66-year-old lawyer with HHT, hypertension, and prior stroke of unknown etiology presents to the emergency department with dyspnea and palpitations. Atrial fibrillation is diagnosed; she is initiated on rate control and admitted for anticoagulation and cardioversion. Her baseline ESS without treatment is 4 to 5, and she uses tranexamic acid with success. Within 6 hours of initiation of heparin infusion, however, she suffers a gushing nosebleed requiring emergent otolaryngology consultation and nasal packing. She undergoes cardioversion during the hospitalization, with recurrence of atrial fibrillation within hours. Her CHA2DS2-VASc (congestive heart failure, hypertension, age, diabetes mellitus, prior stroke or transient ischemic attack or thromboembolism, vascular disease, age, and sex category) score is 6 (yearly stroke risk of 10%-14%). Her cardiology team recommends indefinite therapeutic anticoagulation or procedural management (atrial fibrillation ablation or left atrial appendage closure), which will still require months of periprocedural antithrombotic therapy. Notably, after initiation of aspirin 81 mg daily after her prior stroke, her epistaxis worsened dramatically, resulting in 2 hospital admissions and nasal cautery procedures, and prompting aspirin discontinuation.
Management of HHT-associated bleeding when antithrombotic therapy is indicated
HHT does not impart protection against venous or arterial thromboembolism and indeed appears to increase risk of both, relative to the general population.22,81,82 Therefore, it is critical to treat thromboembolic events and risk as would otherwise be clinically indicated.8 However, approximately one-third to one-half of patients with HHT, even those with mild bleeding at baseline, develop an unacceptable exacerbation of bleeding severity upon initiation of any antithrombotic therapy, including therapeutic-dose anticoagulation, prophylactic dose anticoagulation, and/or antiplatelet therapy (including low-dose aspirin).83-86 In patients with no history of antithrombotic therapy exposure, it should be initiated as indicated under close observation for worsening bleeding. For those in whom it is known to be intolerable, or is found to be intolerable after initiation, bleeding management should be pursued to facilitate indicated antithrombotic therapy.
For antithrombotic-intolerant patients with a required duration of antithrombotic therapy of ∼3 months or less, I refer to an otolaryngologist with experience in local ablative procedures used in HHT for evaluation and local procedural management. For antithrombotic therapy–intolerant patients with a longer or indefinite indicated duration of antithrombotic therapy, I initiate antiangiogenic therapy (bevacizumab or pomalidomide) concurrently with antithrombotic therapy under close observation. Because antiangiogenics usually take 2 to 4 weeks of treatment before achieving therapeutic effect, a local ablative procedure before initiation of dual antithrombotic/antiangiogenic therapy can also be considered.
Similarly, I usually continue antiangiogenic therapy in patients being treated with it who develop a thromboembolic complication while receiving therapy. In the limited published data46,83 and my clinical experience, the hemostatic effect of the therapy allows for uncomplicated antithrombotic treatment in most cases.
Treatment-refractory moderate-to-severe HHT-associated bleeding
A subset of patients with moderate-to-severe HHT-associated bleeding without viable clinical trial options will fail management with both bevacizumab and pomalidomide. In my experience, these patients usually have severe bleeding or red cell transfusion dependency (very severe bleeding). In such situations, I usually first attempt a trial of combined antifibrinolytic plus antiangiogenic therapy. I have not observed any obvious increased thromboembolic risk with this approach, an observation supported by the limited published data.46 If this approach does not achieve acceptable hemostasis, I consider salvage therapies (Table 2). Of note, a prior study of bevacizumab in HHT that evaluated serum drug concentrations found a relationship between low exposure and failure to respond60; because measurement of drug levels is not readily available or interpretable in clinical practice, empiric dose escalation (eg, to 7.5 mg/kg) in nonresponders can be considered, with the caveat that adverse events may be more likely to occur.
Current salvage medical therapies primarily include low-dose oral pazopanib (for epistaxis and/or gastrointestinal bleeding) or parenteral somatostatin analogs (for gastrointestinal bleeding only). Limited observational data suggest effectiveness of low-dose oral pazopanib (50-400 mg daily, in contrast to the oncologic dosing of 800 mg daily), a VEGF receptor tyrosine kinase inhibitor approved to treat solid tumors, in severe HHT-associated bleeding, including patients who have failed bevacizumab.64,65 Like bevacizumab, pazopanib may be more effective to manage gastrointestinal bleeding than epistaxis but can be effective for both. Somatostatin analogs (subcutaneous octreotide or intramuscular long-acting–release octreotide or lanreotide) are parenterally administered peptide hormone analogs that minimize gastrointestinal bleeding via reduction of splanchnic blood flow. Limited observational data suggest effectiveness of somatostatin analogs to reduce gastrointestinal bleeding in HHT, although these data show lower reduction in transfusion requirements and higher rates of discontinuation because of adverse events than studies of bevacizumab in similar HHT populations.66,67
Although major nasal surgery, including a septodermoplasty (essentially a skin graft from the thigh to the nasal mucosa) or nasal closure (Young's procedure; closure of the nares to stop anterior nosebleeds) remain options for patients with severe epistaxis refractory to present medical therapies, these procedures are associated with significant consequences.8 Septodermoplasties must be performed by otolaryngologists with extensive experience in the procedure (a rare commodity) for optimal outcomes, and, even then, hyposmia, hypogeusia, recurrent epistaxis, and particularly unpleasant chronic malodor of the graft are common complications and sloughing off of the graft can occur.87 Nasal closures eliminate the ability to breathe through the nose, often significantly compromise the senses of smell and taste, and do not stop posterior epistaxis or blood gushing down the throat.88 In the modern systemic therapy landscape and given ongoing and upcoming clinical trials, I very rarely refer for consideration of these procedures at present. Most patients in my practice who have failed all available medical therapies opt for repeat local ablative procedures to control epistaxis while awaiting a clinical trial of a novel medical treatment rather than pursuing septodermoplasty or nasal closure.
Conclusions
HHT is a not-so-uncommon inherited bleeding disorder that remains without any US Food and Drug Administration– or European Medicines Agencies–approved treatments. With proper application of off-label systemic therapies and aggressive management of iron deficiency, however, HHT-associated bleeding and its complications can be successfully and durably managed. Development of new systemic therapeutics in HHT is ongoing and will hopefully offer additional safe and efficacious treatment options to reduce morbidity and mortality in HHT as well as enhance HRQoL of these patients.
Acknowledgments
H.A.-S. is funded by the National Institutes of Health, National Heart, Lung, and Blood Institute (grant 1K23HL159313).
Artwork in the visual abstract and Figures 2 and 3 was reproduced and modified from Servier Medical Art (https://smart.servier.com/).
Authorship
Contribution: H.A.-S. was responsible for all aspects of the manuscript, from conception to completion.
Conflict-of-interest disclosure: H.A.-S. reports consulting for Agios, Amgen, Alnylam, Alpine, Sobi, Argenx, Pharmacosmos, and Novartis and received research funding from Agios, Sobi, Amgen, Vaderis, and Novartis.
Correspondence: Hanny Al-Samkari, Division of Hematology Oncology, Massachusetts General Hospital, 55 Fruit St, Bartlett Hall Extension Office 133, Boston, MA 02114; email: hal-samkari@mgh.harvard.edu.